
ampbell Grieg De Morgan (1811-1876) received his medical training at University College, London. At Middlesex Hospital, London, he lectured on forensic medicine in 1841, was appointed assistant surgeon in 1842, became professor of anatomy at the Medical School in 1845; and, in 1853, he was elected to the Royal Society (Grange, J. L. Stanford, and C. A. Stanford, 296). Along with papers on bone growth (1852) and on erysipelas (1860), he is best known for his research on the origin and treatment of cancer and on the senile or cherry angioma, a benign skin lesion commonly known as, "The Campbell De Morgan Spot" (J. Dixon, 334; "De Morgan," 621; Grange, J. L. Stanford, and C. A. Stanford, 296).
As the narratives of Drs. Fell and Pattison have shown, in the mid-Victorian period, conventional and chemical surgery were thought to have been mutually exclusive of each other. But Dr. De Morgan and colleagues, while participants in the 1857 Fell trial at Middlesex Hospital, learned not only that these uniquely-beneficial modalities were compatible with one another, but also that that zinc chloride had anticancer, antiseptic, and hemostatic properties. During the 1860s and early 1870s, he tested the possibility that ablation, in conjunction with chemical therapy, might minimize or preclude surgical complications, might extend periods of remission, and possibly be curative. He would call this a "mixed mode of operating" (The Origin of Cancer, 73)
In 1865, Dr. De Morgan began to sponge zinc chloride lotion into post-surgical wounds. His purpose was to destroy microscopic cancer cells, correctly perceived to be the seeds of recurrence. In 1866, he wrote that the chloride lotion was to be "adopted in the first instance after removal of malignant tumors by the knife" ("On the Use," British and Foreign, 201-02). The mixed-modal system, which was deemed "well worthy of careful trials," incorporated aspects of conventional and of homeopathic medicine. On the one hand, the rapid extirpation of a tumor and surrounding tissues with the knife improved upon the paste which, though effective against small lesions, worked slowly, caused discomfort, and could not keep pace with aggressive disease. On the other hand, unlike the knife, the paste and lotion, applied after the primary tumor had been removed, destroyed residual microscopic cancer and prevented complications (201-02).
Along with the surgical innovation described above, De Morgan contributed important insights to the understanding of cancer pathology. In an 1871-1874 series of papers, delivered at a conference of the Pathological Society of London, he hypothesized that cancer, beginning as a local disease, could spread to the lymphatic system, and then to distant sites in the body (J. M. Grange, J. L. Sanford, and C. A. Sandford 296). The idea that cancer originated as a localized, rather than as a constitutional, called for radical improvements in early diagnosis and treatment.
Continental Innovators
In the 1870s, European physicians reiterated the favorable review that zinc chloride had received at Middlesex Hospital in 1857. Theodor Billroth (1829-1894), of Vienna (probably best known for coining the term Streptoccocus), and Jean Joseph Bougard (1815-1884), of Brussels, for example, had adapted Canquoin's basic formula (No. 1) as a model. Billroth blended equal parts of ZnCl2 with flour to make a strong topical paste to coagulate large tumors. Instead of augmenting his formula with botanicals, however, he turned to a method of delivery, which the French surgeon, Jules Germain Maisonneuve (1809-1897), had invented in 1858. The latter's method had three variants: (1) radiated cauterization: conically-shaped arrows are disposed circularly, at intervals of a centimeter, around the base of the tumor; (2) parallel cauterization: the dried caustic, shaped into pointed lathes, are inserted, not circularly, but “parallel to each other over the whole of its free surface, so that its interior consists of a kind of bundle of caustic arrows, between the interstices of which the tissues are reduced to great thinness, and are rapidly destroyed“; and (3) central cauterization: cut fusiform, the arrows are pressed into the tumor through a surgical puncture or cavity at its center, producing a thick eschar (Maisonneuve 428-9). The method was even used by Dr. J. R. Wolfe, an Ophthalmic Surgeon to the Aberdeen Royal Infirmary who, in May and October of 1867, successfully treated epithelial cancer of the eye with caustic arrows containing carbolic acid (19-20).
Billroth employed Maisonneuve's radiating procedure. After drying his zinc-chloride mixture to the consistency of clay, he cut the material into "small pointed cylinders half a centimeter or more in thickness." Inserting the cones into the tumor, presumably once the tissue had been chemically rendered soft and friable, he implanted them in the neoplasm at varying levels and around the circumference. Each cone, fixed circumferentially ¾ of an inch apart, melted at body temperature, releasing the zinc chloride into the interior of the mass. To control pain, over a four-to-five-hour period, he injected morphine subcutaneously. In twenty-four hours, the tumor routinely was reduced to a "white slough"; and, in five or six days, it separated from the surface. A coagulated ring of tissue at the base of the tumor, intentionally extended from the diseased to the "healthy parts," served as a margin of safety, in case microscopic cancer cells had migrated there. The process was to be repeated if malignant tissue was visible (716).
Dr. Bougard's prescription, the product of nearly 40 years of escharotic research and of 15 years of surgical development, differed from the Fell-Pattison regimens in that it combined four active ingredients, whereas the latter had two (the ZnCl2 and the botanical). The subtitle to Bougard’s 1872 Traitement du Cancer misleadingly asserted that he had invented a new medicine and system; rather, he had simply reinvented Canquoin's original prescription. A powerful amalgam, Dr. Bougard's ointment, delivered via paste or inserted sticks, contained four caustics, amounting to nearly 50% of the mixture: 245 grams of zinc chloride; 1 gram of arsenic; 50 centigrams of corrosive sublimate (mercury chloride), and 5 grams of salt of ammonia. The paste consisted of 60 grams of farina, 60 grams of amidon (starch), and 5 grams of cinnabar (a red mercuric sulfide pigment) (Bougard 66). Bougard's formula and methods, which were conceptually aligned with De Morgan and Maisonneuve, influenced early twentieth-century oncology, a term coined in 1857 (Ohmann-Dumesnil 268; Fordyce 239; "oncology," OED II, 1989).
The Mixed Mode of Operation
De Morgan, who had been studying chemosurgery since the mid-1850s, strongly suspected that prolonged remission depended on the removal of every trace of localized cancer. In 1866, he introduced to Middlesex Hospital the ZnCl2 lotion as a chemotherapeutic adjuvant to surgery. His reasoning was sound: "it is probable that, of the innumerable living cells which are set free [by excision] and deposited in the wound, some will find a nidus in which they will go on developing—they will be scattered like seed, in fact, and grow wherever they find a favourable soil" ("On the Use," The Retrospect, 147-8). This scattering of cells accounted for the appearance of multiple small tumors after a large mass had been extirpated ("On the Use," British and Foreign, 204-05). De Morgan cited as corroborating evidence the work of his colleague, Dr. Charles Hewitt Moore (1821-1870), who had been operating on both skin and mammary tumors, guided by the principal of dissemination. As early as April 1864, he had been managing rodent skin and breast cancer cases through a combination of primary excision and of secondary zinc-chloride applications, as solid and as lotion, to areas where lymph nodes and primary breast tumors had been excised and where recurrence was common (206); routinely, he sponged a "strong lotion" of ZnCl2 "freely over the whole exposed surface" ("On the Use," Retrospect, 151; British and Foreign, 206). Penetrating the surgical margins, and sponged under skin flaps and over muscle, the chloride was intended to destroy "any floating particles of the disease which might adhere to it" ("On the Use," British and Foreign, 206). In cases where conventional surgery either was inadvisable or unsuited to the patient, the dual aspects of the procedure could be reversed: that is, macroscopic, uncoagulated lesions could be surgically removed after the paste had destroyed the bulk of the tumor. In a March 1865 operation, he described a three-stage procedure, involving the chemical destruction of a tumor in a woman’s right breast; the surgical removal of axillary nodes; and the application of moderate strength zinc-chloride solution into the wound (20 grains of ZnCl2 per ounce of water) (206). Post-surgically, according to De Morgan, this particular patient did well: superficial skin sloughed away, pain was minimal, the wound remained aseptic, and healing was rapid. Significantly, from March 1865 to January 1866, she remained in remission. To optimize the combined technique, De Morgan experimented with higher concentrations, 30 and 40 grains of ZnCl2 per fluid ounce, to eradicate extensive disease, and to reduce, as much as possible, the chance of recurrence. De Morgan discovered additional benefits of the caustic. In 1857, he had learned that ZnCl2, though a powerful escharotic, spared healthy tissue, prevented bacterial infections, and promoted rapid healing (210, 213). In 1872, clinical evidence confirmed these observations (The Origin, 65).
By the early 1870s, surgeons had three nonsurgical modes in their anticancer arsenal (66): (1) Galvano- or electric cauterization, a procedure that Albert Theodor Middeldorpf (1824-1868), of Breslau, had developed in 1854; superficial tumors could be cauterized with electricity while the patient was chloroformed (67). Its drawbacks were threefold: battery-power failure, protracted healing, and incomplete eradication of cancerous tissues (67); (2) the circumferential insertion of dissolvable caustic sticks, "to destroy all the intervening tissue . . . isolating and killing the diseased mass" (68). On the negative side, without imaging technology the surgeon, who could only approximate the internal dimensions of the mass, could not guarantee that the zinc chloride had saturated the tumor entirely; and (3) the ZnCl2 paste was slowly applied by hand (68). Despite the inconclusive outcome of the 1857 ZnCl2-S canadensis trial, and although no published evidence suggests that De Morgan and Moore had incorporated S. canadensis into their formula, De Morgan adverted to Dr. Fell's dual-action formula as superior to all others (68-69). Fell's paste had had dramatic results, routinely coagulating tumors, and leaving "a healthy granulating surface." However, despite the immediate effectiveness of the paste, the pace and reach of the therapy remained problematic. As the primary mass was being systematically reduced, new cancerous growth "made progress even before the wound [had] healed"; or the primary cancer that the paste had not, or was unable to, reach kept growing (70). To cover the entire tumor-surface and to accelerate the process, De Morgan advised that zinc chloride paste and lotion be "fully and decisively" administered (70). The radical idea was that the highest tolerable strengths should be employed.
By the early 1870s, cauterization and dissection, combined and robustly applied, had become the state-of-the-art treatment for skin cancers of the head and face (The Origin, 70). Dr. Fell's 1857 trial at Middlesex Hospital, by the 1870s, had inaugurated a major period of research and development in the treatment of rodent and other cancers (71). Canquoin's original formula, and statistics on the management of skin and mammary cancers published in the 1830s, had undergone significant modifications. Even the principal of the hot-iron cautery, which Dr. John Davy had observed in Ceylon 60 years earlier, had been retooled in the 1870s as part of a broader strategy:
The success which has attended the destruction of these rodent ulcers and other malignant growths, at the Middlesex Hospital, should encourage surgeons to attack them more decisively than is usually done. The plan which we have adopted, is to dissect away with the scalpel as much of the diseased structure, especially at its margins, as can be removed; then to cauterize the whole surface and its dissected margins with the hot iron; and lastly, to cover the whole with linen well smeared with the strong chloride of zinc paste—the whole being padded up with cotton wool. These coverings are left undisturbed until they begin to separate; the separation begins at the margins, and the cloths and eschar may be cut away day by day. Below will be found a healthy granular surface which soon cicatrices. [The Origin 71-72]
The "mixed mode of operating," in De Morgan's final analysis, had the potential to lengthen remission time and was incontrovertibly better than "either knife or caustic alone" (73).
The Mixed Mode Enters the Twentieth Century
In 1894, The Johns Hopkins University Hospital surgeon, Dr. William Stewart Halsted (1852-1922), concurrently with Dr. Willy Meyer (1858-1932), of New York, devised what is now known as the radical mastectomy (Donegan 8; C. Eggers). As documented in a 1907 memoir, Halsted had favored a combined therapy. He used a chemical caustic as an adjuvant to the knife: "I am indubitably convinced that the local and [regional] recurrences after incomplete operations, which come as a rule with amazing rapidity when the knife has been used, are, to say the least, relatively late in making their appearance when chemical or actual cauterization has been employed" (18). In his experience, escharotic substances, vigorously and repeatedly applied after surgery, seemed to prevent microscopic cancer cells from migrating to, and growing in, healthy tissue. Dr. Halsted knew that, unless the danger of dissemination was addressed, and if cancer recurred, its "local manifestations . . . were almost invariably deplorable and the prognosis . . . invariably hopeless" (18).
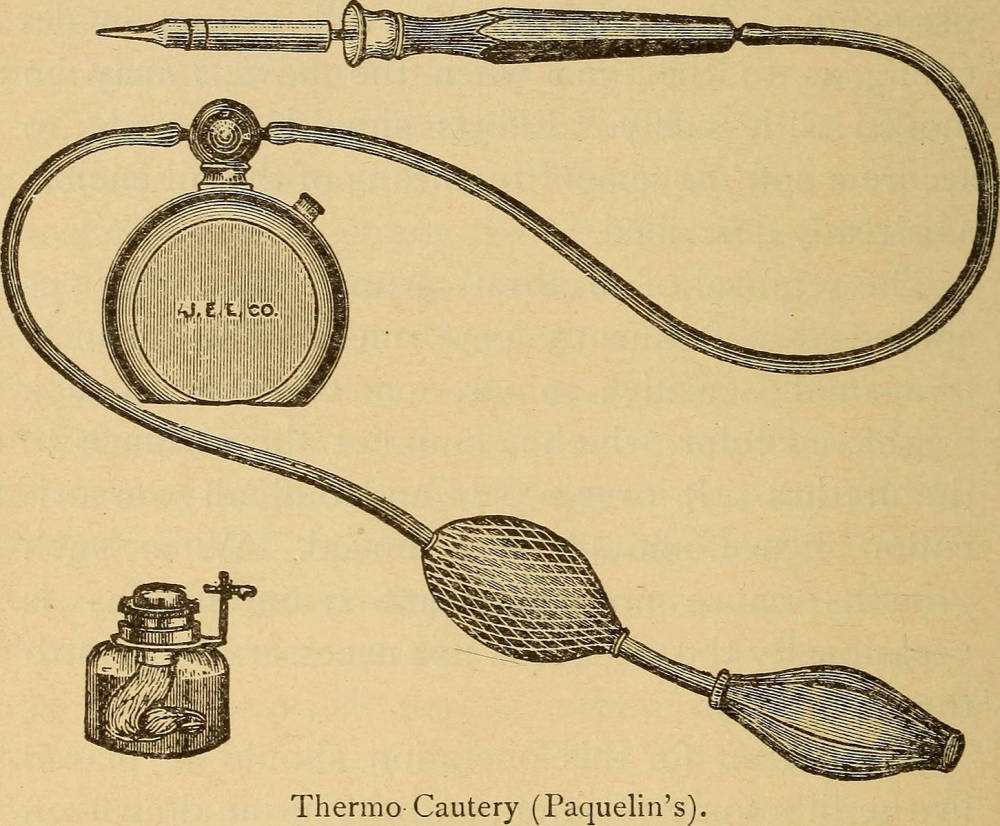
Equipment used in thermocautery from Anna M. Fullerton, Nursing in abdominal surgery and diseases of women (Philadelphia: P. Blakiston & Son, 1893). Courtesy of the Library of Congress and Wikimedia Commons.
Instead of ZnCl2, Dr. Halsted relied on thermal cauterization for exploratory surgery and to guard against the dissemination of malignant cells. Employing an instrument known as Paquelin’s Thermo-Cautery, he was able to destroy exposed parts of a tumor efficiently. Claude-André Paquelin (1836-1905), a student of pharmacology, biochemistry, and medicine, in 1876 had presented his reinvention of the hot-iron cautery to the Acadèmie of Sciences in Paris ("Paquelin's Thermo-Cautery"). As the image shows, the mechanism had a 1.0-1.5-inch long metal tube. Tipped with a half-blunt platinum blade, it was connected to a reservoir containing the coal-tar derivatives, benzene and toluene (called benzoline). As the operator compressed the bulb, air pressure pushed boiling benzoline (heated by a spirit lamp) through the tube, and to the incandescently hot blade; as the exuding benzoline dissolved the tumor on contact, the surgeon, using the blade, debrided the dead tissue (Barwell 388; Sherman 1079; "Benzoline"). Halsted’s use of both cauterization and advanced surgical techniques represented a milestone in the history of cancer therapy.
Bibliography
Barwell, John. "Excision of Lingual Epithelioma by Panquelin's Thermo-Cautère."The British Medical Journal. (Jan.-June 1877). Ed. Ernest Hart. London: Frances Fowke, 1877. 388-92. Google Books. Web. 11 May 2016.
"Benzoline." Medical Dictionary. medical-dictionary. Web. 9 April 2017.
"De Morgan, Campbell Grieg. [Obituary]." The Lancet (22 April 1876) 107.2747: 621. Google Books. Web. 9 April 2017
_____. "On the Use of the Chloride of Zinc in Surgical Operations and Injuries, and especially in Operations for the Removal of Cancerous Tumours." British and Foreign Medico-Chirurgical Review. (January, 1866). London: Savill and Edwards, 1866. 201-216. Internet Archive. Web. 8 July 2015.
_____. "On the Use of Chloride of Zinc in Surgical Operations and Injuries. Surgery: Diseases of the Bones and Joints, Etc." The Retrospect of Medicine, vol. 53. Ed. William Braithwaite. London: Simpkin, Marshall & Company, 1866. 147-162. Google Books. Web. 12 October 2015.
_____. "Correspondence [May, 1868]: Solution of Chloride of Zinc in Surgery." The British Medical Journal, vol. 1. (6 June 1868). Ed. Ernest Hart. London: The British Medical Association, Jan.-June, 1868. 573. Google Books. Web. 18 April 2017.
_____. The Origin of Cancer. Considered with Reference to the Treatment of Disease.. Google Books. London: John Churchill, 1872. Web. 10 March 2017.
_____. "On Cancer." Retrospect of Practical and Medicine and Surgery, vols. 68-69. Eds. W. Braithwaite and James Braithwaite. New York: W. A. Townsend, 1874. 34. Google Books. Web. 7 April 2017.
_____. "Observations on Cancer: Its Pathology, and Its Relation to the Organism and to Other Morbid Growths." The Lancet. (7 April 1874) 1.2636: 323-329. Google Books. Web. 7 April 2017.
Dixon, James. “De Morgan, Campbell Grieg." Dictionary of National Biography, vol. 14. Ed. Leslie Stephen. London: Smith, Elder, & Company, 1888. Internet Archive. Web. 9 April 2017.
Donegan, William L. "History of Breast Cancer." Breast Cancer. Eds. David J. Winchester and David P. Winchester. 2nd edn. Hamilton, Ontario: B. C. Decker, 2006. 1-14. Google Books. Web. 23 February 2017.
Eggers, Carl. "Obituary of Doctor Willy Meyer [1858-1932]." Bulletin of the New York Academy of Medicine. (March, 1932) 8.3: 148-9. National Library of Medicine. Web. 15 April 2017.
Fordyce, John A. "Surgery of the Skin and Its Appendages." Surgery, vol. 6. Ed. William W. Keen. Philadelphia and London: W. B. Saunders Company, 1919. 237-43. Google Books. Web. 10 April 2017.
Grange, John M., John L. Stanford, and Cynthia A. Stanford. "Campbell De Morgan’s 'Observations on Cancer,' and Their Relevance Today." Journal of the Royal Society of Medicine. (June, 2002) 95: 296-299. JRS Sage Publications. Web. 25 November 2015.
Halsted, William Stewart. "The Results of Operations for the Cure of Cancer of the Breast Performed at the Johns Hopkins Hospital from June 1889 to January 1894." Annals of Surgery. (1894) 20.5: 497-555. National Library of Medicine. Web. 29 February 2016.
Maisonneuve, Jules Germain François. "On a New Mode of Cauterization." The Medical Times and Gazette, vol. 17. London: John Churchill, 1858. 428-9. Google Books. Web. 29 April 2017.
Meyer, Willy. "An Improved Method of the Radical Operation for Carcinoma of the Breast." Medical Record (15 December 1894). New York: Trow Directory, 1894. Internet Archive. Web. 7 April 2017.
Middeldorpf, Albert Theodor. Die Galvanokaustik, ein Beitrag zur Operativen Medizin. Breslau: Max & Company, 1854.
Ohmann-Dumesnil, Amant H. Handbook of Dermatology: For the Use of Students. 2nd ed. St. Louis: Quarterly Atlas Company, 1894. Google Books. Web. 10 April 2017.
"Oncology." Compact Edition of The Oxford English Dictionary. 2 vols. Oxford: At the Clarendon Press, 1972. 1989.
"Paquelin's Thermo-Cautery." Nursing in Abdominal Surgery and Diseases of Women. By Anna Martha Fullerton. Philadelphia: P. Blakiston, Son, & Company, 1893. 250 (Fig. 66). Google Books. Web. 22 April 2017.
Sherman, Samuel. "Further Observations on the Technique of an Efficient Procedure for the Removal and Cure of Superficial Malignant Growths." The Journal of Cutaneous Diseases. (October, 1910) 28.10: 1079. JAMA Network. Web. 12 April 2017.
Wolfe, J. R. "The Removal of Cancer by Caustic Arrows and Carbolic Acid." The Retrospect of Medicine, vol. 58. Eds. W. Braithwaite and James Braithwaite. London: Simpkin, Marshall, and Co., 1869. Google Books. Web. 29 April 2017.
Last modified 6 May 2021